
Assessing data quality and the variability of source data verification auditing methods in clinical research settings | Semantic Scholar
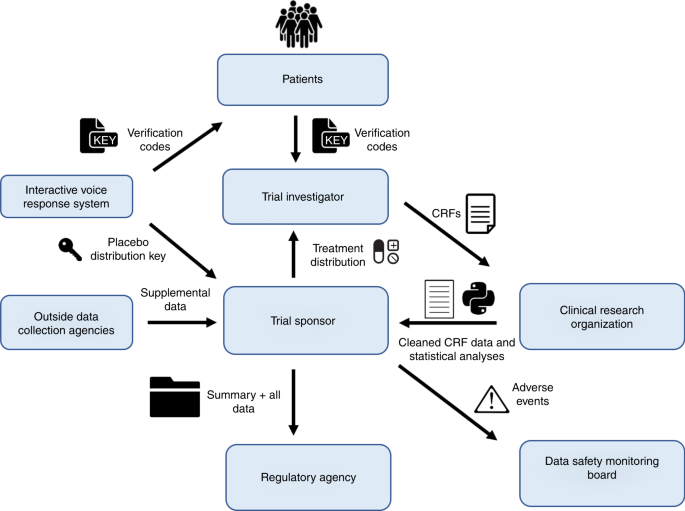
Prototype of running clinical trials in an untrustworthy environment using blockchain | Nature Communications

High Point Clinical Trials Center Partners with Verified Clinical Trials to Ensure Participant Safety and Data Quality - Verified Clinical Trials
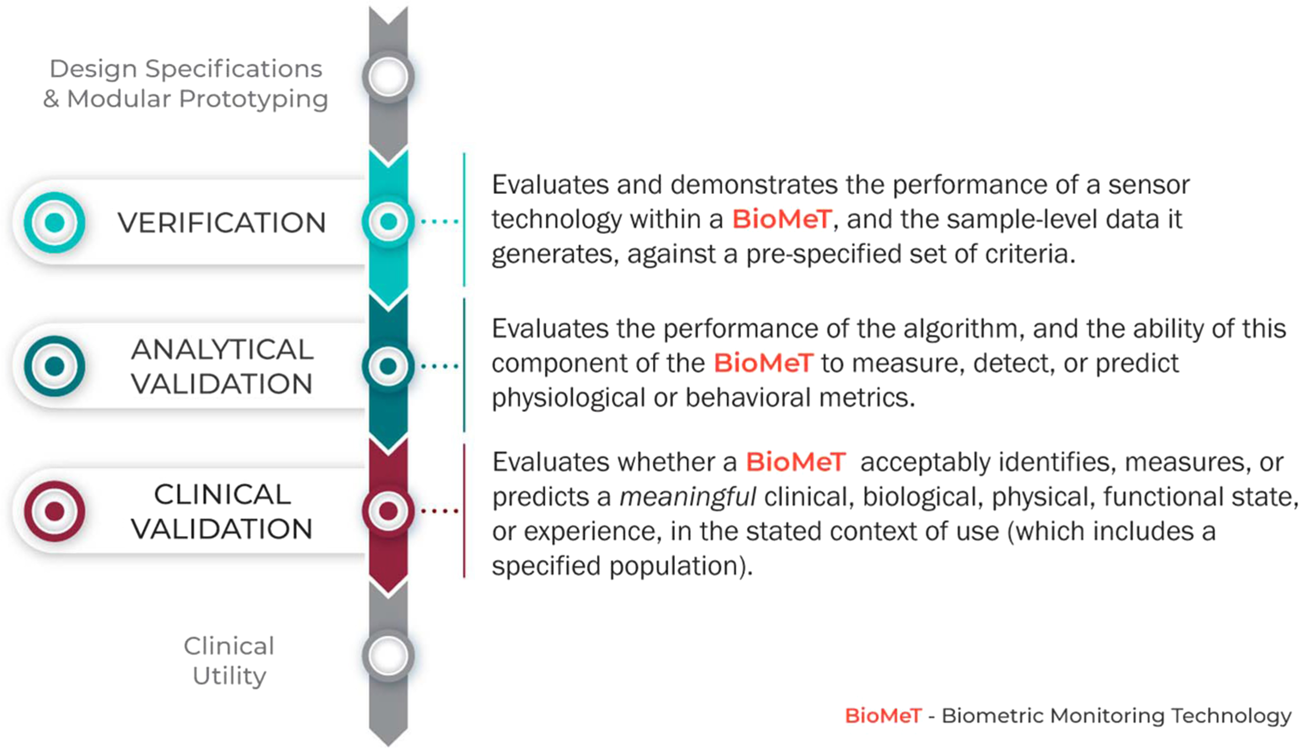
Verification, analytical validation, and clinical validation (V3): the foundation of determining fit-for-purpose for Biometric Monitoring Technologies (BioMeTs) | npj Digital Medicine

Data Integrity in Global Clinical Trials: Discussions From Joint US Food and Drug Administration and UK Medicines and Healthcare Products Regulatory Agency Good Clinical Practice Workshop - Khin - 2020 - Clinical

Research Centers of America Adopts Verified Clinical Trials Biometrics Research Subject Database Registry to Prevent Duplicate Enrollment in Clinical Trials

Source Data Review (SDR) vs. Source Data Verification (SDV) – A Site Monitoring Best Practices Update - Life Science Training Institute

The Role of Source Data Verification (SDV) and Source Data Review (SDR) in Driving Clinical Trial Data Quality - Medidata Solutions

Pillar Clinical Research Selects Verified Clinical Trials As Exclusive Provider To Reduce Professional Subjects From Duplicate Enrollment In Their Clinical Trials.














