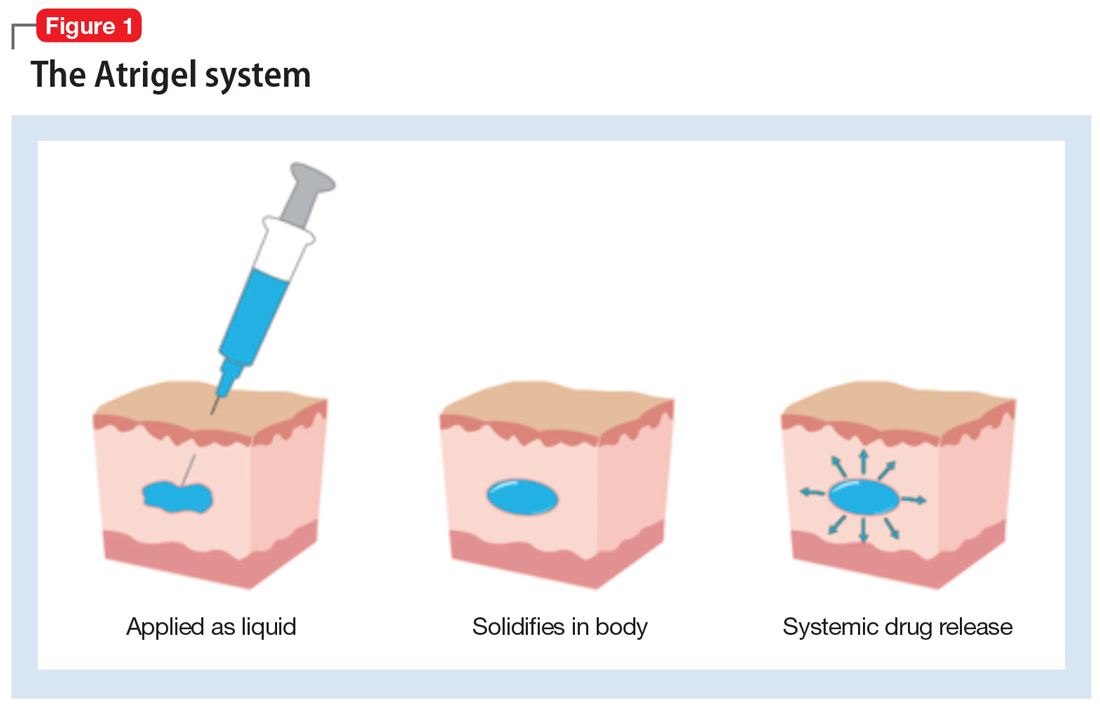
Teva and MedinCell looking to enter LAI schizophrenia market - Pharma Technology Focus | Issue 103 | February 2021

Yo-Bio on Twitter: "$TEVA will compete in the risperidone Extended-Release market with $JNJ (risperdal consta- every 2 weeks, INTRAMUSCULAR) and $INDV (PERSERIS- every 4 weeks, subcutaneous). if Teva regimen and efficacy will
PROPOSAL FOR THE ADDITION OF RISPERIDONE LONG-ACTING INJECTION AND PALIPERIDONE PALMITATE 1-MONTH LONG-ACTING INJECTION FOR THE

Teva and MedinCell Announce FDA Acceptance of New Drug Application for TV-46000/mdc-IRM as a Treatment for Patients with Schizophrenia | Business Wire

FDA declines to approve Teva-MedinCell's risperidone injection for treating schizophrenia | Seeking Alpha
Teva and MedinCell looking to enter LAI schizophrenia market - Pharma Technology Focus | Issue 103 | February 2021

Teva and MedinCell Announce Positive Results for Registration Trial of Investigational Extended-Release Subcutaneous Injectable Risperidone for Patients with Schizophrenia | Business Wire
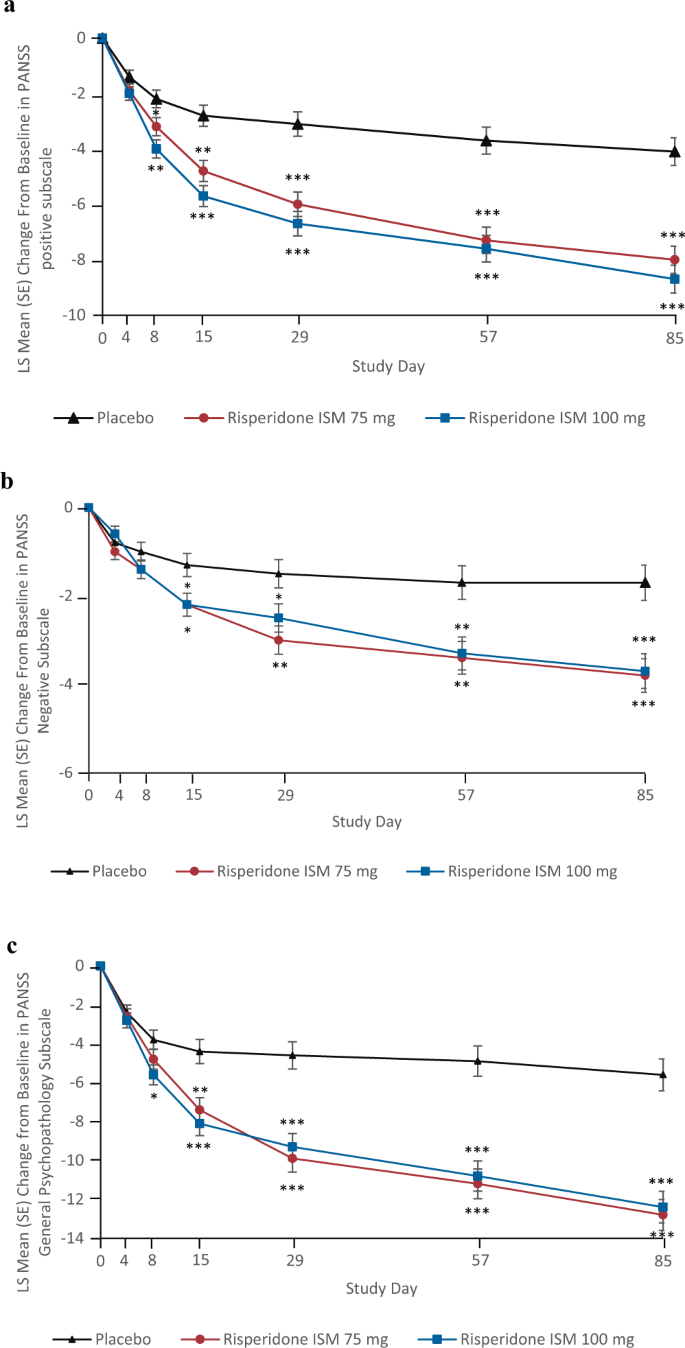
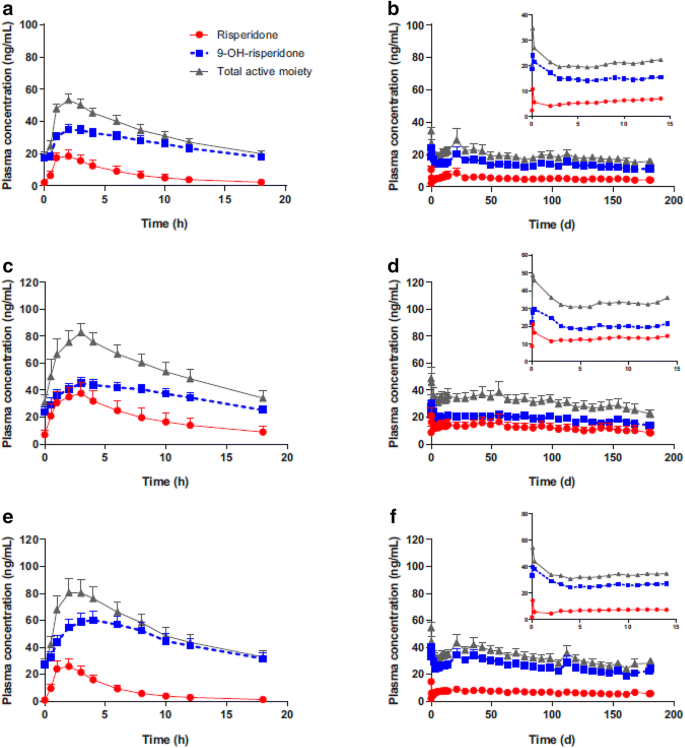
Efficacy and safety of once-monthly Risperidone ISM® in schizophrenic patients with an acute exacerbation | Schizophrenia

Teva and MedinCell Announce Positive Results for Registration Trial of Investigational Extended-Release Subcutaneous Injectable Risperidone for Patients with Schizophrenia | Business Wire

Teva and MedinCell Announce Positive Results for Registration Trial of Investigational Extended-Release Subcutaneous Injectable Risperidone for Patients with Schizophrenia | Business Wire








/cloudfront-us-east-2.images.arcpublishing.com/reuters/Q3V76PIRQBKVFCT4F2KKAGPMDE.jpg)