Calculate the biling point of a solution containing 0.456 g of comphor (molar mass=152 g mol^(-1)) dossoved in 31.4 g of acetone(b.p.= 329.45 K). Given that the molecular elevation constant per 100 g of solvent is 17.2 K.

Resistance change of MWCNT BP immersed in acetone (a), ethanol (b) and... | Download Scientific Diagram
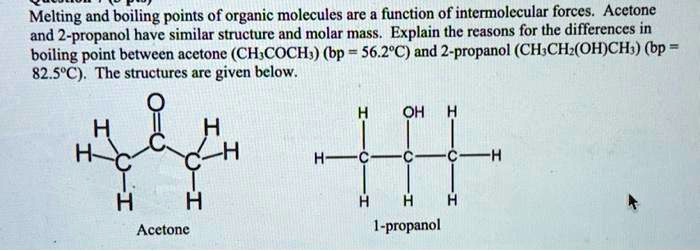
SOLVED: Melting and boiling points of organic molecules are function of intermolecular forces: Acetone and 2-propanol have similar structure and molar mass. Explain the reasons for the differences in boiling - point

a) STEM image of FL-BP acetone sheets. (b) STEM image of a selected... | Download Scientific Diagram

SOLVED: (bp=5o"C): The Acetone (bp-56"C) and n-hexane (bp-70*C) form a binary minimum boiling azeotropei 'distilled composition of the azeotrope is 60% acetone and 40% n-hexane: Suppose that you fractioeiallyadi 100.0 mL sample

SOLVED: Consider the following list of liquids and their boiling points: ether, bp 35'C acetone bp = 568C cyclohexane, bp 80PC water, bp = 100PC Which liquid is predicted to have '

![PDF] Separation of Water-Acetone Mixture Using Suitable Entrainer (Simulation) | Semantic Scholar PDF] Separation of Water-Acetone Mixture Using Suitable Entrainer (Simulation) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3b24cb3fdaba29b0182f3943cf35a512430d1196/29-Table3-2-1.png)